Description
HUMOG 150 for Sale in USA without prescription
Menotropins For Injection U.S.P.
Menotropin (also called human menopausal gonadotropin or hMG) is a hormonally active medication for the treatment of fertility disturbances. Frequently the plural is used as the medication is a mixture of gonadotropins. Menotropins are extracted from the urine of postmenopausal women.
HUMOG (Human Menopausal Gonadotropin) is a hormonal substance containing FSH and LH in a ratio of 1:1. In the female, HUMOG stimulates both the growth and the maturation of follicles, it induces an increase in the oestrogen levels and a proliferation of the endometrium. In the male, HUMOG stimulates the spermatogenesis by acting on the production of the androgen binding protein in the seminiferous tubules of the Sertoli cells.
INDICATIONS
Women:
HUMOG and subsequently HUCOG (Human Chorionic Gonadotrophin) are indicated for the induction of ovulation in the amenorrhoeic patient or anovulatory women with regular or irregular cycles.
Men:
HUMOG with concomitant HUCOG therapy is indicated for the stimulation of spermatogenesis in men who have primary or secondary Hypogonadotrophic hypogonadism.
DESCRIPTION AND USAGE
Urine of postmenopausal women reflects the hypergonadotropic state of menopause-levels of follicle stimulating hormone (FSH) and luteinizing hormone (LH) are high – and contain a mixture of these gonadotropins. Other protein substances may be present, including small amounts of human chorionic gonadotropin (hCG). In 1949 Piero Donini found a relatively simple method to extract gonadotropins from urine of postmenopausal women. Menotropins were successfully introduced into clinical use by Bruno Lunenfeld in 1961.
Menotropin preparations are designed for use in selected women where they stimulate the ovaries to mature follicles, thus making them more fertile. They are administered by typically daily injection, intramuscularly or subcutaneously, for about ten days under close supervision to adjust dose and duration of therapy. They can also be used in hypogonadal men to stimulate sperm production.
Human urinary-derived menotropin preparations are exposed to the theoretical risk of infection from menopausal donors of urine. Nevertheless, the failure to irrefutably demonstrate infectivity following intracerebral inoculation with urine from transmissible spongiform encephalopathy (TSE)-infected hosts suggests that the risk associated with products derived from urine is merely theoretical.
Recombinant gonadotropins have to a large degree replaced hMG in fertility treatments. The recombinant process allows for the production of pure FSH or LH not “contaminated” by other proteins that may be present after urinary extraction. While some head-on studies seem not to suggest that “pure FSH” gives better results than hMG, others claim that recombinant FSH is more efficient and reduces costs. A Cochrane Collaboration analysis did not reveal major differences in clinical outcomes when comparing urinary versus recombinant FSH.
The Practice Committee of the American Society for Reproductive Medicine reported: “Compared with earlier crude animal extracts, modern highly purified urinary and recombinant gonadotropin products have clearly superior quality, specific activity, and performance. There are no confirmed differences in safety, purity, or clinical efficacy among the various available urinary or recombinant gonadotropin products”.
DOSAGE AND ADMINISTRATION
HUMOG is given by intramuscular injection.
Reconstitute powder of vial in 1ml of Sodium Chloride Injection I.P. provided in the pack immediately prior to use. Up to 5 vials of HUMOG may be Reconstituted in 1 ml of Sodium Chloride Injection. Reconstituted solution should be used immediately after preparation. Any unused portion of solution should be discarded.
Women:
The object is to develop a single matured Graffian follicle with individually tailored doses of HUMOG over several days and to give HUCOG to release the ovum. Follicular development is judged by the concentration of oestrogen, measured in blood or urine. Clinical assessment of the response including pelvic examination and cervical mucus studies should also be performed. HUMOG administration should continue until an adequate oestrogen level is achieved.
If the oestrogen level is less than either 180nmol/24 hr. (50μg/24 hr) for tested urinary oestrogen or 1100pmol/L (300pg/ml) for plasma 17ß-oestradiol, follicular development may be inadequate. Conversely, if the levels are higher than either 514nmol/24 hr (140μg/24 hr) for total urinary oestrogens or 3000pmol/L (800pg/ml) for plasma 17ß-oestradiol, there is an increased risk of ovarian hyperstimulation and HUCOG should be withheld. The optimal time for HUCOG administration is the day of the urinary oestrogen peak or the day after the plasma 17ß-oestradiol peak. In the anovulatory patient the stimulated follicles will not liberate ova spontaneously. Follicular rupture had to be achieved by injecting HUCOG which stimulates the normal surge of LH at ovulation.
If the patient wishes to conceive, she is recommended to have coitus on the day when HUCOG is given and on the following day. The dose of HUMOG required to evoke the desired response is critical and varies both from patient to patient and in the same patient at different times. Monitoring by hormones assay is therefore essential
Two dosage schedules may be employed:
Schedule 1 : Alternate day therapy
Three equal doses of HUMOG are given on alternate days. In menstruating woman the initial dose of HUMOG should be given on day 7, 8 or 9 of the cycle. A single dose of HUCOG 10000 I.U. is given one week after the first injection of HUMOG, provided the clinical and biochemical responses are adequate and not excessive.
Schedule 2 : Daily therapy
Daily injections of HUMOG are given until an adequate response is achieved. This is judged on the basis of daily oestrogen determinations. In the absence of a response, the dose of HUMOG may be increased or the course abandoned. A single HUCOG injection of 10000 I.U. is administered 24-28 hours after the last dose of HUMOG. Schedule 2 is most commonly used.
Men:
Treatment should begin with HUCOG 2000 I.U. 2-3 times a week to produce evidence of adequate masculinisation. If the response to HUCOG is only androgenic, HUMOG (1 vial 3 times a week) and HUCOG 2000 I.U. (twice a week) are required to be administered.
CONTRA-INDICATIONS AND WARNINGS
Women:
HUMOG therapy is precluded when an effective response cannot be obtained e.g. Ovarian dysgenesis, Absence of uterus, Premature menopause, Tubular occlusion.
Men:
Patients with elevated endogenous FSH levels indicative of primary testicular failure are usually unresponsive to HUMOG and HUCOG therapy.
Appropriate treatment should first be given for hypothyroidism, adrenocortical deficiency, hyperprolactinaemia or pituitary tumour. An acceptable semen analysis should be avaliable before HUMOG treatment.
Adherence to the recommended dosage and monitoring schedules will minimise the possibility of ovarian hyperstimulation. Excessive oestrogenic response to HUMOG do not generally give rise to significant side effects unless HUCOG is given to induce ovulation. Hormone assays will detect an excessive oestrogen response to HUMOG and HUCOG. In such cases HUMOG administration should be withheld. The incidence of multiple births following HUMOG / HUCOG therapy has been variously reported between 10% and 40%. However, the majority of multiple conceptions are twins. Pregnancy wastes by abortion is higher than in a normal population but comparable with the rates in woman with other fertility problems. The risks of congenital abnormalities are not increased by HUMOG.
SIDE EFFECTS
In the female, a local reaction at the injection site, fever and arthralgia have been observed in rare cases.
In the male, a combined treatment with HUMOG and HUCOG may cause gynecomastia.
STORAGE
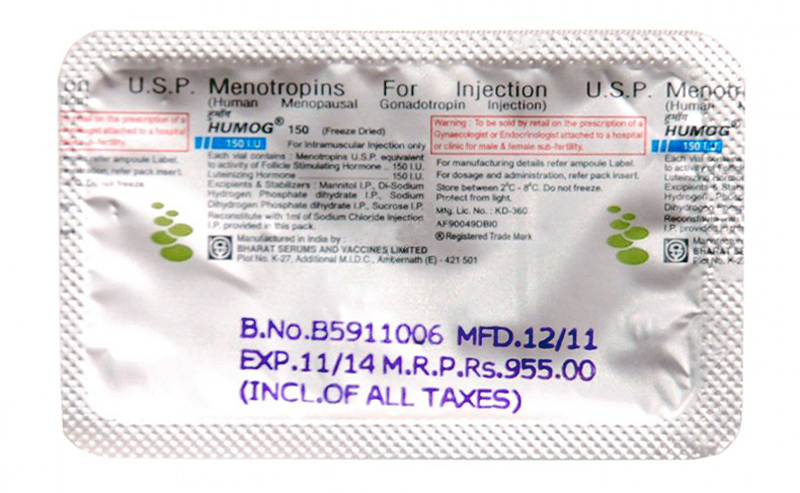
Vials of HUMOG should be stored between 20C-80C. Do not freeze. Protect from light.
Reconstituted solution of HUMOG should be used immediately after preparation. Discard any unused portion.
PRESENTATION
HUMOG is supplied in vial containing sterile, freeze dried white powder having 75 I.U. / 150 I.U. activity of each FSH and LH . Each vial is accompanied by an ampoule containing 1ml of Sodium Chloride Injection I.P.


Reviews
There are no reviews yet.